- Síndromes de compressão vascular
- Tem alguma pergunta a fazer?
- Checklist vascular compression syndromes
- Particularidades musculoesqueléticas da puberdade feminina
- Lordose / cruz oca – causa de numerosas síndromes de compressão abdominal
- A síndrome do Quebra-nozes é um nome errado.
- May-Thurner-Konstellation /May-Thurner-Syndrom/Cockett’s syndrome/Vena iliaca-Kompressionssyndrom
- Síndrome da Linha Média (Síndrome de Congestionamento da Linha Média)
- Síndrome de congestão pélvica (congestão dos órgãos pélvicos)
- Compressão de Truncus coeliacus / Síndrome de Dunbar / MALS / Síndrome de Ligamentum arcuatum
- Síndrome de Wilkie / Síndrome de Arteria-mesentério-superior
- Quantificação de síndromes de compressão vascular com a técnica PixelFlux
- Neuralgia pudendal em síndromes de compressão vascular
- Tratamento das síndromes de compressão vascular
- Síndromes de compressão vascular recentemente descobertas
- Ultrassom Doppler colorido funcional – como eu o entendo
- Medição do fluxo sanguíneo – Método PixelFlux
- Competência
- Colaboração científica
- Cookie Policy
- Cookie Policy (EU)

A rare variant of Wilkie syndrome
A rare variant of Wilkie syndrome
(published on 20.01.2024 on the 41st birthday of Prof. Dr. rer. nat. Jakob Scholbach, who developed the first version of the PixelFlux software as a pupil at the Ostwald-Gymnasium in Leipzig)
Wilkie syndrome (SMA syndrome, superior mesenteric artery syndrome) is a compression of the duodenum by the superior mesenteric artery when it crosses the aorta. This arterial clamp constricts the duodenum to such an extent that shortly after ingestion of food, the transportation of food immediately behind the stomach outlet (after 6 cm) stops and the food is pressed against this clamp, causing severe pain. The duodenum often expands to a diameter of 3 or more centimeters – which causes the pain – and the food spills back into the stomach at the end of a peristaltic contraction wave, often even into the mouth.
As a result, patients vomit, regurgitate, the stomach acid flows back, they have severe wave-like pain in the right upper abdomen and on the line connecting the navel and sternum when the futile contraction waves are thrown back at the arterial clamp. Finally, patients may suffer a potentially life-threatening loss of weight.
Unfortunately, this clinical picture, especially in young women and girls, is diagnosed as anorexia nervosa (psychogenic eating disorder) without a thorough physical examination. The consequences for the patients are devastating. They are accused of having severe psychological conflicts as the cause of their complaints, the pain is not believed, the complaints are only imagined and the vomiting is induced arbitrarily in order to lose weight.
In my opinion, the diagnosis of anorexia without ruling out vascular compression is a major mistake.
I would now like to present a previously undescribed variant of this clinical picture.
The very slim young patient (BMI minimum 15.5 – currently 18.7 under tube feeding) had a hypermobile Ehlers-Danlos syndrome.
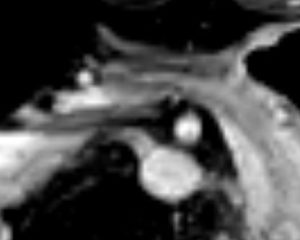
 After feeding, the filling stomach pushed the superior mesenteric artery further to the right to a distance of 23 mm from the aortic axis. Now the filling stomach pushed the superior mesenteric artery to a minimal distance of 7.12 mm to the anterior edge of the lumbar vertebral body. A space of about 3 mm remained for the duodenum. This resulted in closure of the duodenum. The right-sided portion of the duodenum expanded to 4 to over 5 cm. The high peristaltic pressure manifests itself in the fact that the contour of the duodenum rears up in a shoulder shape at the superior mesenteric artery (arrows). The stomach also has hardly any space (5.95 mm) in the median line, whereby the pancreas is compressed to 6.38 mm. (Such patients sometimes have impaired pancreatic function or slightly elevated lipase levels in the blood). The distance between the spine and the inner abdominal wall is only 2.16 cm, which points to the high degree of lordosis as the cause of this and the patient’s accompanying vascular compression syndromes (lordogenetic renal vein compression – often incorrectly referred to as “nutcracker syndrome” , May-Thurner syndrome and ligamentum arcuatum syndrome).
After feeding, the filling stomach pushed the superior mesenteric artery further to the right to a distance of 23 mm from the aortic axis. Now the filling stomach pushed the superior mesenteric artery to a minimal distance of 7.12 mm to the anterior edge of the lumbar vertebral body. A space of about 3 mm remained for the duodenum. This resulted in closure of the duodenum. The right-sided portion of the duodenum expanded to 4 to over 5 cm. The high peristaltic pressure manifests itself in the fact that the contour of the duodenum rears up in a shoulder shape at the superior mesenteric artery (arrows). The stomach also has hardly any space (5.95 mm) in the median line, whereby the pancreas is compressed to 6.38 mm. (Such patients sometimes have impaired pancreatic function or slightly elevated lipase levels in the blood). The distance between the spine and the inner abdominal wall is only 2.16 cm, which points to the high degree of lordosis as the cause of this and the patient’s accompanying vascular compression syndromes (lordogenetic renal vein compression – often incorrectly referred to as “nutcracker syndrome” , May-Thurner syndrome and ligamentum arcuatum syndrome).
 It is therefore not surprising that the aorta, which is often depicted in the middle of the spine in anatomical textbooks, has deviated 1.70 cm to the left of the midline. The superior mesenteric artery, on the other hand, has deviated 4.81 mm to the right of the midline. The situation can best be described as follows: the spine squeezes the upper abdominal organs against the inner abdominal wall, causing the arteries to deviate to the side, but the structures that run horizontally across the spine, such as the duodenum, pancreas and stomach, are highly compressed, which leads to an emptying disorder of the stomach and duodenum.
It is therefore not surprising that the aorta, which is often depicted in the middle of the spine in anatomical textbooks, has deviated 1.70 cm to the left of the midline. The superior mesenteric artery, on the other hand, has deviated 4.81 mm to the right of the midline. The situation can best be described as follows: the spine squeezes the upper abdominal organs against the inner abdominal wall, causing the arteries to deviate to the side, but the structures that run horizontally across the spine, such as the duodenum, pancreas and stomach, are highly compressed, which leads to an emptying disorder of the stomach and duodenum.
As long as the patient was fasting, the distance between the superior mesenteric artery (SMA) and the spine was 13.8 mm, whereby the artery had already deviated 5.3 mm to the right of the aortic axis (see following figure):
The superior mesenteric artery was displaced far to the right after feeding. The aorta was located on the left anterior curvature of the spine. Both vessels therefore described a wide angle in the horizontal plane.
In Wilkie syndrome and all other vascular compression syndromes in the abdominal cavity, increased lordosis of the lumbar spine (hollow back) plays a decisive role in the disease.
This was also the case in this patient. The pronounced forward curvature of the spine meant that there was only a gap of approx. 2 cm in front of the spine for the abdominal organs. As a result, the aorta, the stomach and the superior mesenteric artery did not have enough space to lie in a sagittal plane, as is usually depicted in standard anatomical textbooks.
The confined space had led to the duodenum being trapped between the superior intestinal artery and the spine instead of between the superior intestinal artery and the aorta, as is usually the case in Wilkie’s syndrome. The peristaltic wave was unable to elevate the superior iliac artery far enough against the abdominal wall to allow the food to pass through the horizontal portion of the duodenum unimpeded. The symptoms corresponded to those of a classic Wilkie syndrome.
It is important not to overlook those variants in which the classic anatomical constellation is not present, but in which the functional consequences and thus the clinical picture represent a considerable impairment for the patient. It is particularly impressive how the position of the upper abdominal organs changes due to the additional volume required by the filling stomach. This can produce completely different anatomical relationships than those usually seen on a fasting image.
For example, this fasting CT of the patient shows a completely different, “normal” topography of the ASM and aorta, namely in a sagittal plane:
This frequent observation once again emphasizes the need for a functional examination and close observation of the interaction of the upper abdominal organs during the functional test. Assuming an upright posture can also cause considerable changes in the position of the organs. It is not uncommon for the renal blood flow to be impaired after eating or while standing, with the increasing congestion of blood in the collateral circulation then triggering significant symptoms in the legs and pelvis during food intake and upright posture.
In the patient, the blood flow in both kidneys collapsed significantly when standing. However, even food intake causes a decrease in blood flow to the left and right kidneys when lying down.
 The following diagram shows that the peripheral blood flow in the right kidney drops from 53% in the fasting state to 44% after food intake. This is a clear indication of the increasing outflow resistance of the right renal vein, obviously due to its compression by the massively dilated duodenum.
The following diagram shows that the peripheral blood flow in the right kidney drops from 53% in the fasting state to 44% after food intake. This is a clear indication of the increasing outflow resistance of the right renal vein, obviously due to its compression by the massively dilated duodenum.
A quantitative assessment of the blood flow to the organs with the PixelFlux software, which was first developed 24 years ago by my son, Jakob Scholbach, is indispensable for the assessment of the manifold correlations of the disturbed organ function of various abdominal organs and provoking factors such as changes in body position and food intake.