- Studio medico
- Sindromi di compressione vascolare
- Avete domande?
- Lista di controllo delle sindromi di compressione vascolare
- Particolarità muscoloscheletriche della pubertà femminile
- Lordosi/croce cava – causa di numerose sindromi di compressione addominale
- La sindrome dello schiaccianoci è un termine improprio.
- Sindrome di May-Thurner /May-Thurner-Syndrome/Cockett’s syndrome/Sindrome da compressione della vena iliaca
- Sindrome della linea mediana (Sindrome di congestione della linea mediana)
- Sindrome da congestione pelvica (congestione degli organi pelvici)
- Compressione del celiaco del tronco / Sindrome di Dunbar / MALS / Sindrome di Ligamentum arcuatum arcuatum
- Sindrome di Wilkie / Arteria-mesenterica-sindrome superiore
- Quantificazione delle sindromi di compressione vascolare con la tecnica PixelFlux
- Nevralgia pudendale nelle sindromi di compressione vascolare
- Trattamento delle sindromi di compressione vascolare
- Sindromi da compressione vascolare recentemente scoperte
- Ultrasuoni Doppler a colori funzionali – come faccio io
- Collaborazione scientifica
- Cookie Policy
- Cookie Policy (EU)

Venous congestion of the spinal cord may be a potential contributor to the development of paraplegia in patients with spinal muscular atrophy type III (Kugelberg-Welander disease)
Preface
I have specialised in diagnosing and treating vascular compression for over 35 years. During this time, I have seen many patients with leg weakness, some of whom were so severely affected that they could no longer walk and were either bedridden or required a wheelchair. All of these patients had spinal congestion due to a shortcut between the pressurised and compressed left renal vein and the spinal cord. This vessel was first identified by French anatomists and is thus named in French “tronc réno-rachidien”. This translates in English to “reno-spinal trunk “. After successfully decompressing the left renal vein and the left common iliac vein, which are often compressed simultaneously in patients with left renal vein compression, these patients regained muscle strength in their legs and were able to walk again.
This is why I saw similarities between these patients and those with spinal muscular atrophy which affect the leg muscles earlier and more severely than the muscles of the upper body in patients with SMA II, III and IV.
Despite nearly all these disorders being caused by a defect in the SMN-1 gene whichis active in all neurons, I wondered why they start in the legs and only later affect the arms and torso.
However, the most severe forms with very early onset , SMA 0 and SMA I, do not follow this pattern: in SMA 0 patients present with facial paralysis at birth, whereas in SMA I, facial muscles are initially spared but are later involved and the muscles of the entire body are affected from the beginning .
Having treated many patients with vascular compression syndromes and spinal congestion, which result in leg weakness, I considered whether spinal congestion might contribute to the severity and progression of spinal muscular atrophy.
Congestion occurs when venous blood exerts pressure on an organ due to a combination of maintained arterial influx and obstructed venous outflow, or additional venous blood entering a vascular territory when the organ becomes part of a collateral network of veins.
Within the central nervous system, we can formally distinguish between congestion of the brain and congestion of the spine. Congestion of the brain primarily affects the brain itself and the cranial nerves that innervate the muscles of the face, neck and shoulders. Due to the extensive communication between the spinal cord and skull at the foramen magnum at the base of the skull, the pressure within these structures may shift depending on body posture.
Spinal congestion would affect the spinal nerves, depending on the hydrostatic pressure of the congesting fluid — in this case, venous blood. If the spine is upright, the pressure is higher in the lumbar and lower thoracic regions than in the upper thoracic and cervical regions. However, if the spine is horizontal, the pressure is distributed evenly throughout the spine.
From the perspective of congestion of the central nervous system, damage to the muscles of the head, neck, and shoulders would occur when the brain is congested. This is exactly what is seen in SMA type 0, which starts in the embryo. In contrast to SMA type I, which starts in the first months of life and spares the facial muscles, SMA type 0 affects the facial muscles from the beginning and all the other muscles too.
In SMA I, congestion of the CNS affects the cranial nerves less than in SMA 0. Under the assumption of the theory presented here, the posture of the baby is crucial.
The baby either lies horizontally most of the time (while sleeping) or is held by the parents with its head up and its legs down. If congestion would be at work, this could explain why cranial nerves, and thus facial muscles, are primarily unaffected in babies with SMA I, and why the effect on the arms is stronger and occurs earlier in SMA I than in types II to IV.
Since the fetus usually lies head down during the mother’s upright posture (sitting or standing), congestion of the CNS affects the upper body, including the cranial nerves, more strongly than in SMA types that start later in life, when patients are already upright for most of the day.
This is the case in SMA types II , III and IV. They start in childhood or adulthood. Then CNS congestion would affect the caudal parts of the spinal cord more strongly than the cranial parts and the brain due to the high hydrostatic pressure in the lumbar and lower thoracic spine while being upright when the spine is congested .
If congestion plays a role in the development of progressive muscle paralysis in SMA patients, the various patterns of muscle damage from SMA 0 to SMA IV would fit with the idea that the highest congestive pressure is in the part of the CNS that is closer to the Earth’s surface. Gravitation works in a vertical line, so the pressure in a fluid column is highest at the bottom and lowest at the top. Assuming that higher congestive pressure leads to greater neuronal damage, the fact that embryos and newborns are more affected in the upper body, while SMA patients with later-onset show stronger muscle damage innervated from the lower (caudal) part of the spine, supports this hydrostatic theory of an additional factor influencing the course of SMA.
95% of 4 types of spinal muscular atrophy are caused by a deficiency of SMN (survival motor neuron protein ) encoded by the SMN-1 or SMN-2 gene.
These 4 types of SMA differ in their age of onset of clinical symptoms and severity of its course and outcome.
SMA I starts in newborn and babies and is often fatal in early childhood. The lower limbs are generally weaker than the upper limbs.
SMA II starts around the age of 6-8 months with primary involvement of the legs. The children achieve the ability to sit only with assistance.
Patients with SMA III learn to walk more than 10 steps and maintain this ability into adulthood when the weakness starts in the legs as well.
SMA IV is a mild disease with little functional impact probably due to an increased number of SMN-2 copies.
Interestingly, among the 5% of SMA subtypes which are not caused by SMN-1 deficiency and not rarely also start at the lower limbs.
Case report
Here, I report on a 64-year-old female patient with four copies of SMN2 and progressive weakness of the lower limbs, which required the use of a wheelchair intermittently from the age of 48 and now nearly constantly.
Medical history
The patient could only rise from a sitting position with support; an assistant would pull her belt above the sacral bone while she used her arms to support her torso. She would place her hands on the table in front of the chair as soon as she could extend her knees. From this position, she could then stand up by suddenly contracting her trunk muscles, which moved the spine into an erect posture in a whiplike fashion.
Apart from her leg paralysis, the patient has no other complaints. She reports no headaches or back pain. She shows signs of hypermobile joints and loose connective tissue: both her elbow and knee joints can be hyperextended, and she was previously able to touch the floor with her palms while standing. This gives a Beighton score of 5/9, which suggests hypermobility syndrome. At a younger age, she could put her legs behind her neck. The patient has no striae distensae or dilated subcutaneous vessels on her abdomen. She wears compression stockings due to chronic oedema in both legs. So far, this has been attributed to the immobilisation of the patient.
Clinical examination
Abdominal palpation reveals no localised pressure pain or pathological resistance. Auscultation is also unremarkable, with no vascular bruits and normal peristalsis. The abdomen is protruding due to extreme lordosis resulting from increasing gluteal muscle weakness. However, the patient reports having had increased lordosis even before significant paralysis of her legs set in.
Quantitative colour Doppler sonography
A colour Doppler sonography with quantification of the tissue perfusion of the uterus and the kidneys as well as flow volume measurements in both internal iliac veins is undertaken.
The uterus is massively enlarged and shows a substantial increase of venous blood vessels pointing to a severe pelvic congestion which so far was not felt by the patient.
The maximum tissue perfusion of the pelvic organs measured under standardised conditions (cm/s * cm² of the perfused area of the ROI /cm² of the ROI – normal < 0.05) is:
Organ Mean value (%) Maximum value (%)
====================================
uterus 678.0% 1532.0%
The quantification of the tissue perfusion of the uterus with the PixelFlux technique reveals a massive congestion of the uterus.
The flow volume measurement in both internal iliac veins discovers a striking asymmetry of the pelvic venous drainage. The left internal iliac vein is transporting 96 mL/min cephalad and the vessel is slim whereas the right one is much wider transporting 960 mL/min cephalad. Thus instead of an equal drainage with 50% transport volume on each side the left side is transporting only 9% whereas the right side has taken over 91%.
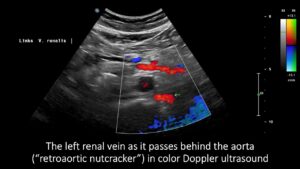
The cause of the severe pelvic congestion is the significant compression of the left common iliac vein at the top of its path over the promontory. This compression does not occur directly at May-Thurner’s point, where the vein crosses the right iliac artery. While the vein has a sagittal width of 11 mm to the left of the sacrum, where blood flow is below the detection threshold, the funnel-shaped narrowing of the vein as it ascends to the promontory leads to intermittent blood flow in the compressed section immediately ventral to the fourth lumbar vertebral body along the median line. Here, the vein has a sagittal width of less than 1 mm, resulting in blood flowing at speeds of up to 132 cm/s. However, blood flow can only be detected during inspiration. This is therefore a severe case of pelvic venous outflow obstruction and May–Thurner syndrome, with leg oedema being the only clinical manifestation.
A detailed examination reveals a left ascending lumbar vein with cranially directed blood flow, carrying a volume of approximately 72 ml/min, which is likely to contribute to spinal congestion.
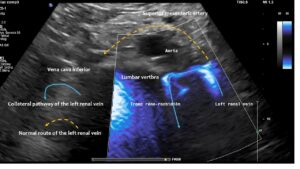
Rather than being located between the superior aorta 100, the left renal vein curves around the left lateral circumference of the spine, draining blood to the spine via a strong vessel known as tronc réno-rachidièn . Additionally, a very narrow retroaortic second left renal vein is present.
These images, taken from the front of the patient, show the tronc réno-rachidièn. This vessel runs in a U-turn along the left side of the lumbar spine towards the back, entering the intervertebral foramen and thus putting pressure on the spinal canal. They demonstrate that the preaortic portion of left renal vein (yellow dashed arrow) does not carry any blood, meaning that all of the left renal venous blood must be diverted via the tronc réno-rachidièn. The other natural collateral pathway, the left ovarian vein, is less significant here. In this patient ( as in many arteries with left renal vein compression) , the collateralisation via the left ovarian vein towards the pelvic venous circulation is obstructed by compression of the left common iliac vein. This combination is very common and requires simultaneous treatment of both the left renal vein and the left common iliac vein in one operation, by shielding both with a PTFE shield.
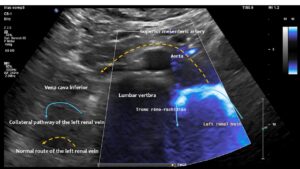
Blood within the tronc réno-rachidièn is modulated in a pulsatile manner, demonstrating rhythmic compression of the left renal vein between the aorta and superior mesenteric artery. This compression is so pronounced that no blood flow is visible ventral to the aorta only the pressure wave is transmitted towards the collateral vessel.
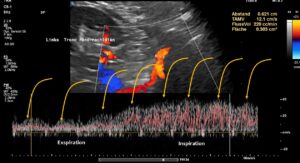 This image demonstrates the transfer of the pulsatile pressure of the aorta and the superior mesenteric artery towards the tronc réno-rachidièn (yellow arrows). Both arteries completely squeeze the final portion of the left renal vein obstructing the entire blood flow, but the pressure is rhythmically transferred towards the spinal canal and thus the spinal cord. Not rarely affected patients develop headaches and migraines which was not the case in this patient.
This image demonstrates the transfer of the pulsatile pressure of the aorta and the superior mesenteric artery towards the tronc réno-rachidièn (yellow arrows). Both arteries completely squeeze the final portion of the left renal vein obstructing the entire blood flow, but the pressure is rhythmically transferred towards the spinal canal and thus the spinal cord. Not rarely affected patients develop headaches and migraines which was not the case in this patient.
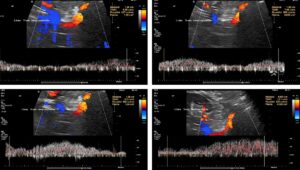 These images show the respiratory variability of volume transport within the tronc réno-rachidèn.
These images show the respiratory variability of volume transport within the tronc réno-rachidèn.
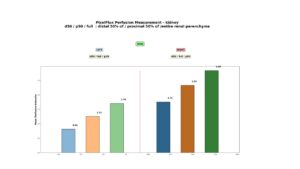
The following diagrams show that the PixelFlux measurement of blood flow in the renal parenchyma indicates a significant reduction in blood flow to the left kidney. This is due to severe venous outflow obstruction via the subtotally compressed left renal veins, both pre- and retro-aortic. The left kidney receives only 53% of the perfusion volume of the right kidney. Peripheral parenchymal perfusion is also significantly lower on the left than on the right, reflecting congestion pressure in the spinal canal. This prevents the tronc réno-rachidien from draining more blood from the congested left renal vein, thereby relieving the kidney. Conversely, this demonstrates that the substantial pressure exerted on the spinal canal, and consequently on the spinal cord, may be a significant contributing factor to paraparesis in SMA III.
Diagram of perfusion measurement of the renal parenchyma using the PixelFlux technique
The columns show the perfusion intensity in cm/s, calculated as the perfusion velocity [cm/s] of all coloured pixels multiplied by the area of all coloured pixels [cm²] divided by the area of all pixels in the entire region of interest [cm²].
The following videos of the patient’s MR venography confirm and illustrate the ultrasound findings. However, ultrasound is more informative than MRI, as it can measure the direction and volume of blood flow in the collateral vessels and compare renal perfusion on both sides. From this, the pressure within the spinal canal can be deduced, which may exacerbate and accelerate the paralysis of the legs.
In a second patient with spinal muscular atrophy, significant venous compression with inadequate collateral circulation and evidence of spinal cord congestion were also observed. The following video summary describes the sonographic findings and explains the pathophysiological effect of inadequate collateral circulation, which can be detected using PixelFlux measurement of renal parenchymal perfusion.
Discussion
This is the first description of a spinal congestion in a patient with progressive paralysis of both legs which so far was attributed to a spinal muscular atrophy type III . It is well known however that in patients without a genetic defect of the SMN-1 gene can develop a paraparesis. This was described in a paper by Frantz, Aboulker, Küss and Jardin in 1977. They describe the development of paraplegia into patient’s with obstruction of the left renal vein and development of a tall in the tronc réno-rachidièn and subsequent congestion of the spinal cord. These authors successfully treated the paraplegia by ligation of the tronc réno-rachidièn. This however caused renal damage in 1 of their to patient’s.
They describe the development of paraplegia in patients with obstruction of the left renal vein and the development of a renal-spinal trunk and subsequent congestion of the spinal cord. These authors successfully treated the paraplegia by ligating the renal-spinal trunk. However, this led to kidney damage in one of their two patients. In a later published paper, they achieved significantly better results. However, they never eliminated the compression of the left renal vein, which leads to higher risks when ligating the renal spinal trunk if there are no other sufficient collaterals. Nevertheless, the connection to the compression of the left renal vein was well known to French authors..Highlighted Les afferences veineuses reno-rachidiennes dans les myelopathies - Aboulker, Jardin - Unknown copy
Another French working group was investigating venous congestion in myelopathy by venograms and confirms our observation that numerous venous vessel compressions often occur simultaneously and contribute to congestion of the spinal cord with corresponding clinical consequences. I am therefore making this extremely informative work available here.
Highlighted aubin-et-al-1975-phlébographie-cavo-rachidienne-dans-les-myelopathies-d-origine-veineuse copyWe (Thomas Scholbach) made the diagnosis and (Wilhelm Sandmann) provided the treatment for multiple patients with a tronc réno-rachidien, due to severe compression of the left renal vein and subsequent progressive paraplegia, necessitating the use of a wheelchair. This avoided the renal damage reported by Frantz et al. in 1977 and also restored the patients’ ability to walk. They were able to walk again and perform other normal daily activities, including dancing and surfing, after the compressed veins were decompressed. The French group succeded in 21 cases of myelopathies and 9 cases of syringomyelia in ligating the tronc rèno-rachidièn.iHighlighted Responsabilité de la Veine Rénale Gauche et de la Veine Cave Inférieure Dans Certains cas de Myélites et de Syringomyélie Intéret de la Ligature du Tronc Veineux Réno-Rachidien
It therefore seemed probable that, in patients with a genetic defect causing muscular weakness, additional spinal cord congestion may contribute to paraplegia.
From a haemodynamic and gravitational medicine perspective, it is understandable that spinal cord congestion primarily affects the more caudal portions due to the human body’s upright position during the daytime in SMA II – IV. This increases the pressure in the caudal parts of the spinal canal, thereby affecting the caudal spinal cord. The exact molecular mechanism of this damage needs to be explored further, but the prompt clinical effect of compressing the spinal cord by reducing the additional venous influx from the collaterals of the compressed abdominal and pelvic veins clearly proves the causal relationship between loss of muscle function and spinal congestion in non-SMA paraplegics.
Therefore, it is advisable to consider an additional haemodynamic cause of paraplegia in patients with a genetic diagnosis of spinal muscular atrophy. This could help to slow the progression of the disease by alleviating spinal congestion through surgery to decompress the left renal vein and the left common iliac vein.