- संवहनी संपीड़न सिंड्रोम
- क्या आपका कोई सवाल है?
- चेकलिस्ट संवहनी संपीड़न सिंड्रोम
- महिला यौवन की मस्कुलोस्केलेटल विशेषताएं
- लॉर्डोसिस – कई उदर संपीड़न सिंड्रोम का कारण
- नटक्रैकर सिंड्रोम एक मिथ्या नाम है
- मे-थर्नर नक्षत्र / मे-थर्नर सिंड्रोम / कॉकटेल सिंड्रोम / वेना इलियाक संपीड़न सिंड्रोम
- पेल्विन्स कंजेशन सिंड्रोम (श्रोणि अंगों का जमाव)
- ट्रंकस कोएलियाकस संपीड़न / डनबर सिंड्रोम / MALS / लिगामेंट आर्किकेट सिंड्रोम
- विल्की सिंड्रोम / बेहतर मेसेन्टेरिक धमनी सिंड्रोम
- PixelFlux तकनीक का उपयोग करके संवहनी संपीड़न सिंड्रोम की मात्रा
- संवहनी संपीड़न सिंड्रोम में पुदाल तंत्रिकाशूल
- संवहनी संपीड़न सिंड्रोम का उपचार
- संवहनी संपीड़न सिंड्रोम जो मैंने हाल ही में खोजा है
- Kaleidoscope of instructive cases
- कार्यात्मक रंग डॉपलर अल्ट्रासाउंड – जैसा कि मैं इसे समझता हूं
- अनुसंधान
- विशेषज्ञता
- वैज्ञानिक सहयोग
- Cookie Policy
- Cookie Policy (EU)

Inauguration of the global volumetric brain perfusion measurement-a gateway for understanding of neurological symptoms
Content
Increased intracranial pressure. 3
4D-PixelFlux global volumetric brain perfusion measurement. 3
Introduction
It is a truism that the normal function of any organ depends on a sufficient supply with blood. However, the proper evaluation of blood supply is still in its infancy. In daily medical routine the measurement of blood supply only involves arterial influx [1]. The venous side of the circulation is less well investigated. However, since blood needs to circulate from the heart to the peripheral organs and back again, the function of the veins — returning blood to the heart — is equally important to the arterial influx.
The damage to an organ resulting from a disturbed blood supply can be evaluated relatively easily by testing its function. This can be done through laboratory tests, such as measuring blood levels of enzymes and metabolic products, and functional tests, such as performance tests, EEGs, ECGs and scintigraphy.
From a patient’s perspective, impaired function manifests as a symptom.
Reduced arterial blood flow to an organ leads to ischaemia, a lack of oxygen and nutrients, and impaired organ function.
However, disturbed and reduced venous outflow results in congestion of the organ, causing increased blood pressure and blood stasis within the organ. This creates oedema, whereby fluid leaks from pressurised blood vessels into the surrounding tissue between the cells of the organ. The oedema increases the distance between capillaries and cells, thus reducing the exchange of vital substances for the survival and function of the organ. The result is a further disturbance to the organ’s function, or even its demise. In most cases, the impaired function is the main issue, accompanied by sensations of pressure, fullness, and pain resulting from increased pressure inside and around the blood vessels as a consequence of the oedema.
Cerebral congestion
In the case of the brain, disturbed venous outflow is experienced as pressure, fullness and headaches, as well as disturbances to brain function, such as impaired vision, cognitive impairment, learning difficulties, memory loss, spatial disorientation [2], fatigue, positional disorientation, dysphasia, retro-orbital headaches, nosebleeds, vomiting, CSF leaks, photopsia, tinnitus and brain fog.[3]
Such cerebral congestion is difficult to detect, as there has previously been no practical method of quantifying global venous outflow from the brain in an outpatient setting. However, this issue has now been resolved with the introduction of the 4D-PixelFlux global volumetric brain perfusion measurement technique.
The normal organs’ blood supply while resting is given in the table below
( from GUYTON AND HALL TEXTBOOK OF MEDICAL PHYSIOLOGY, 14. Ed. 2021, Table 17-1 p 206: in https://cuvas.edu.pk/cuvas_libraries/ebooks/guyton-and-hall-textbook-of-medical-physiology-14th-edition_043155.pdf ) [4]
| Organ / Tissue | % of Cardiac Output | Blood Flow (ml/min) | Blood Flow (ml/min/100g tissue) |
| Liver Global | 27 | 1350 | 95 |
| Kidneys | 22 | 1100 | 360 |
| Liver Portal | 21 | 1050 | |
| Muscle (inactive state) | 15 | 750 | 4 |
| Brain | 14 | 700 | 50 |
| Liver Arterial | 6 | 300 | |
| Skin (cool weather) | 6 | 300 | 3 |
| Bone | 5 | 250 | 3 |
| Heart | 4 | 200 | 70 |
| Other tissues | 3,5 | 175 | |
| Bronchi | 2 | 100 | 25 |
| Thyroid gland | 1 | 50 | 160 |
| Adrenal glands | 0,5 | 25 | 300 |
| Total | 100 | 5000 |
Arterial cerebral perfusion runs via the internal carotid arteries (72–80%), the vertebral arteries (20–28%), and the spinal arteries (with negligible flow volumes, which are negligible for brain supply).
Venous cerebral perfusion is realised by the internal jugular veins, the vertebral veins, and the external and internal spinal veins (epidural and perispinal), which have the following flow volumes:
| Vessel System | Supine Flow (Total) | Upright/Sitting Flow (Total) |
| Internal Jugular Veins | ~740 mL/min | ~200 mL/min |
| Vertebral Veins | ~40–70 mL/min | ~150–250+ mL/min |
| Vertebral Venous Plexus* | Minimal | Dominant (Remaining Outflow) |
With only about 2% of the body weight the brain receives 15% of the cardiac output.
Increased intracranial pressure
Many cerebral and neurological symptoms are related to increased intracranial pressure or disturbed perfusion of certain parts of the brain. The rise of intracranial pressure is a consequence of increasing volume of intracranial structures while the intracranial space remains constant. The intracranial space of an adult is roughly 1500 mL (female) to 1600 mL (male) while the brain volumes are 1300/1400 mL respectively. The fact that the volume difference is only 200 ml, compared to blood flow volumes of 700 ml/min on both the arterial and venous sides, highlights the importance of fine-tuning arterial and venous brain perfusion to prevent a rapid increase in intracranial pressure. If a venous obstruction occurs, the only possible shift in volume is the displacement of cerebrospinal fluid. Its intracranial volume is approximately 180 ml in both sexes, and it is not easily shifted due to its partial location in the intracranial ventricles. The remaining peripheral CSF can be expelled from the skull into spinal reservoirs, but this creates little additional space for trapped venous blood.
The consequences of increased intracranial pressure are wide ranging from dizziness and headaches to vomiting, visual disturbances, dysfunction of the cranial nerves and autonomic disturbances [5] and in the emergency setting even brain herniation, ischemia and death [6].
Chronically increased intracranial pressure is less easily suspected when the main symptoms are fluctuating and less severe, such as various kinds of headaches, creeping vision loss, palsy of the sixth cranial nerve causing double vision, pulsatile tinnitus and dizziness, which nevertheless interfere with normal daily activities.
Idiopathic chronic intracranial hypertension is a disease that mainly affects women and is of unknown origin. Chronic fatigue syndrome may be an early stage of increased intracranial pressure [7].
Precise measurement of intracranial pressure requires invasive techniques, which are not feasible in an outpatient setting. However, cerebral MRI and CT scans can provide clues when the peripheral CSF is reduced and the ventricles are narrower or wider than normal.
4D-PixelFlux global volumetric brain perfusion measurement
I developed a novel, non-invasive functional and quantitative method which is suitable for outpatient evaluation and does not require injection of contrast media-the global arterial and venous brain perfusion measurement with the 4D- PixelFlux technique.
This method was introduced on the basis that constant intracranial pressure requires balanced arterial inflow and venous outflow to and from the skull. If the venous outflow is restricted, or if additional venous volume enters the skull via the epidural and perispinal veins, then the intracranial pressure will become elevated. The intracranial pressure may increase further if this imbalance is unstable.
Colour Doppler ultrasound is the preferred method to evaluate the cranial blood supply since it is harmless, widely available, requires no preparation and is non-invasive. So far however only approximated arterial flow volumes could be calculated with a simple two-dimensional technique while reliable venous flow measurements were out of reach.
For arteries a good approximation was achieved since the flow volume can be estimated by multiplying the mean flow velocity which is measured during the colour Doppler examination of the vertebral and carotid arteries with the transsectional area of the vessel. This area is calculated by simply measuring the largest diameter of the vessel and calculating the area by the circle-area formula. this is not quite exact but a workable approximation.
However, this method is completely misleading in veins. This is due to the strong variability of the cross-sectional area of the vein, which rarely resembles a circle, and the uneven distribution of flow velocities inside the large cervical veins. Flow velocities and area also change significantly due to backpressure from the heart, the effect of respiration, and the neighbouring arteries and muscles. In addition, veins are heavily affected by changes in head position.
To overcome these limitations, we developed the 4D-PixelFlux technique. This enables precise, pixel-wise measurement of flow velocities and the transsectional area of all cervical blood vessels including veins for the first time, taking into account their ever-changing shape and course during head movements.
Moreover, this technique for the first time can calculate potential discrepancies of arterial influx via the carotid and vertebral arteries and the venous outflow via the internal jugular veins and vertebral veins.
This is of the utmost importance in cases of vascular compression syndrome in the abdomen, pelvis, thorax and neck. The spinal canal is an alternative route for many of these compression syndromes, as the vessels within it are protected from local compression. Blood that enters the spinal canal rises within the spine and can enter the skull via the greater foramen at the base of the skull, which contains the medulla oblongata.
With the simultaneous determination of the true flow volumes within both carotid arteries, both vertebral arteries, both internal jugular veins on both vertebral veins we can measure the influx from the spinal canal into the skull contributing to the increased intracranial pressure. There is no relevant arterial influx via the greater foramen. The volume flow difference of the main cervical arteries and veins is thus the additional spinal flow volume.
If such a situation exists the body usually responds with increased outflow via the internal jugular veins. There is little additional transport capacity of the vertebral veins since they are surrounded by the bony foramina in the of the lateral processi of the cervical vertebrae.
If an influx into the spinal canal exists due to venous compression syndromes, the cervical veins are often compressed simultaneously, since increased lumbar lordosis is the cause of all compression syndromes, and this is often accompanied by increased cervical lordosis. This results in compression of the internal jugular vein between the carotid arteries, the omohyoid muscle, and the sternocleidomastoid muscle in the middle of the neck. In such a situation the adaptation to additional spinal influx may cause increased intracranial pressure which can be demonstrated by the flow acceleration at the compression site of the internal jugular vein.
Simultaneously evaluating this pressure gradient and flow volume discrepancy is key to understanding many of the patients’ cerebral symptoms resulting from increased intracranial pressure, which conventional techniques fail to detect.
Case presentation
I present the case of an 18-year-old female patient who has suffered from left-sided migraines, extreme chronic fatigue, exhaustion, weakness in all four limbs, brain fog, hot flushes, dizziness, and abdominal pain for the past two years. Notable additional symptoms included severe menstrual pain, weight loss of 5 kg, nausea, and an inability to stand for more than five minutes.
She displayed clear clinical signs of hypermobility disorder (hypermobile Ehlers-Danlos syndrome). Functional sonographic diagnostics revealed severe pelvic congestion syndrome as a consequence of May–Thurner syndrome combined with left renal vein compression, resulting in extensive collateralisation via a tronc réno-rachidièn towards the spinal canal. Additionally, a developing median arcuate ligament syndrome and severe right-sided orthostatic nephroptosis were identified. An orthostatic syncope occurred during the examination.
Compression of the left internal jugular vein between the sternocleidomastoid muscle and the common carotid artery produced only slight localised flow acceleration, from 24 to 47 cm/s. By contrast, the wider right internal jugular vein exhibited much stronger flow acceleration despite less severe compression at the same cervical level: from 53 to 131 cm/s.Such a discrepancy cannot be explained using conventional colour Doppler techniques. The usual finding is that the faster the flow acceleration, the greater the narrowing of the blood vessel. In this case, however, the opposite was true.
The resolution of this dilemma and explanation of the left-sided headaches, chronic fatigue, brain fog, dizziness was only found with the global arterial and venous 4D- Pixel Flux volume flow measurements of all brain supplying cervical blood vessels.
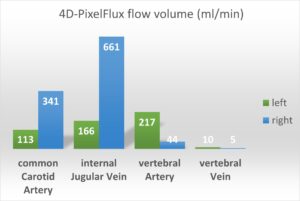
A 4D volume flow measurement is carried out with the patient lying on her back without a pillow, with head in a neutral position. The measurements are outlined in the diagram below. A comparison with normal flow volumes, as indicated below the diagram, shows that the internal jugular veins have different transport capacities. The left vein transports less than 50% of the normal mid-range volume, whereas the right vein has compensatory hyperperfusion, balancing out the loss on the left side. This corresponds to the patient’s left-sided migraine.
The arterial flow volume on the left is 330 ml/min, whereas on the right it is 385 ml/min, which is not a relevant difference.
The overall arterial flow volume is therefore 715 ml/min. The venous outflow from the skull is 167 mL/min on the left side and 665 mL/min on the right side since the vertebral vein cannot compensate for the reduced perfusion volume of the internal jugular vein on the left side.
The overall venous drainage volume is 832 mL/min.
This differs from the arterial influx by 127 mL/min, which corresponds to a venous overload from the spinal canal of 16% of cerebral perfusion.
These measurements show that, on the left side, symmetric arterial brain perfusion meets outflow resistance, necessitating a volume shift within the skull from left to right in order to drain venous blood. This corresponds to the pressurisation of the left intracranial hemisphere, which explains the patient’s left-sided migraine. This is due to the tronc réno-rachidièn, which pumps 270–590 ml/min of blood from the compressed left renal vein towards the spinal canal. The varying volumes are due to changes in body posture; the patient was evaluated in both the horizontal and standing positions. While the cervical 4D- PixelFlux flow volume measurements do not show the total flow volume over time of the tronc réno-rachidièn pointing to an exit of parts of the volume on its way up to the skull via the intervertebral foramina, the influx is strong enough to produce the aforementioned symptoms and flow accelerations. It is now clear that the significant acceleration in flow despite the only slight compression of the right internal jugular vein is due to its volume overload, since the left internal jugular vein is unable to provide the necessary transport capacity.
| Vessel | Arteries | Arteries | Veins | Veins |
| left | right | left | right | |
| Common carotid | 113 | 341 | ||
| Internal jugular | 166 | 661 | ||
| Vertebral artery | 217 | 44 | ||
| Vertebral vein | 10 | 5 | ||
| Sum | 330 | 385 | 176 | 666 |
| Global sum | 715 | 842 | ||
| Venous surplus | 127 |
The common carotid artery supplies the brain and with branches of the external carotid artery the face, the thyroid gland and the neck muscles. However, the flow volume of the external carotid artery is unaffected by impaired venous drainage from the skull, so it can be considered constant. In contrast, the flow volume of the internal carotid artery is affected by intracranial pressure, and thus by venous outflow obstruction from the skull. Therefore, it is justifiable to use flow volume measurements of the common carotid artery in this context instead of the internal carotid artery.
The mean flow volumes of the internal jugular veins in healthy individuals have been determined as follows:
Left internal jugular vein: mean 258.6 ml/min (SD 133.8 ml/min)
Right internal jugular vein: mean 460.8 ml/min (SD 193.8 ml/min) [8]
Common carotid arteries 470±120 ml/min [9]
The video above describes the 4D- PixelFlux volumetric flow measurement Illustrated on 3 different vessels of the same patient
Conclusion
Global 4D-PixelFlux volume flow measurement can describe the effect of spinal congestion on venous blood flow across the skull. This is important since many patients with compression of the internal jugular vein (mostly on the left side) are also affected by abdominal and pelvic venous compression syndromes, such as nutcracker syndrome, lordogenic compression of the left renal vein, and May–Thurner syndrome (lordogenic compression of the left common iliac vein). In such situations, the spinal canal acts as an escape route for venous blood to return to the heart from the congested veins. Common connections include the left common iliac vein via the left ascending lumbar vein and the hemi-azygos vein towards the epidural plexus, and the left renal vein via the tronc réno-rachidièn, towards the epidural plexus. Epidural plexus congestion then forces blood into the draining veins of the spine , including the lumbar, thoracic and cervical segmental veins, as well as into the skull via the epidural plexus, which connects spinal veins with the intracranial venous system across the greater occipital foramen.
If, in addition to this, the outflow is obstructed by a compression of the internal jugular vein , venous congestion may develop in the brainstem and other cranial areas. To reliably assess the volume load, the ensuing intracranial pressure rise, and the volume that needs to be shifted from the obstructed side to the contralateral side, the global 4D PixelFlux volume flow measurement is a long awaited solution to fill a painful void in existing imaging procedures.
Literature
- Michael Peters, A., The precise physiological definition of tissue perfusion and clearance measured from imaging. European Journal of Nuclear Medicine and Molecular Imaging, 2018. 45(7): p. 1139–1141.
- Wei, H., et al., Cerebral venous congestion alters brain metabolite profiles, impairing cognitive function. Journal of Cerebral Blood Flow & Metabolism, 2023. 43(11): p. 1857–1872.
- Arun, A., et al., A proposed framework for cerebral venous congestion. The Neuroradiology Journal, 2022. 35(1): p. 94–111.
- Hall, J.E., Hall M.E., GUYTON AND HALL TEXTBOOK OF MEDICAL PHYSIOLOGY, 14. Ed. 2021. 2021: Elsevier.
- Dunn, L.T., RAISED INTRACRANIAL PRESSURE. Journal of Neurology, Neurosurgery & Psychiatry, 2002. 73(suppl 1): p. i23–i27.
- Olaru, C., S. Langberg, and N.S. McCoin, A review of the clinical presentation, causes, and diagnostic evaluation of increased intracranial pressure in the emergency department. Western Journal of Emergency Medicine, 2024. 25(6): p. 1003.
- Higgins, J.N.P. and J.D. Pickard, A paradigm for chronic fatigue syndrome: caught between idiopathic intracranial hypertension and spontaneous intracranial hypotension; caused by cranial venous outflow obstruction. Fatigue, 2021. 9(3): p. 139–147.
- Feng, W., et al., Quantitative flow measurements in the internal jugular veins of multiple sclerosis patients using magnetic resonance imaging. Reviews on recent clinical trials, 2012. 7(2): p. 117–126.
- Schöning, M., J. Walter, and P. Scheel, Estimation of cerebral blood flow through color duplex sonography of the carotid and vertebral arteries in healthy adults. Stroke, 1994. 25(1): p. 17–22.